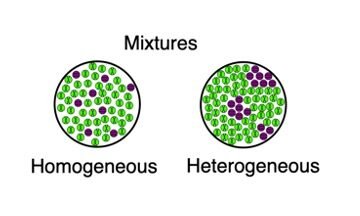
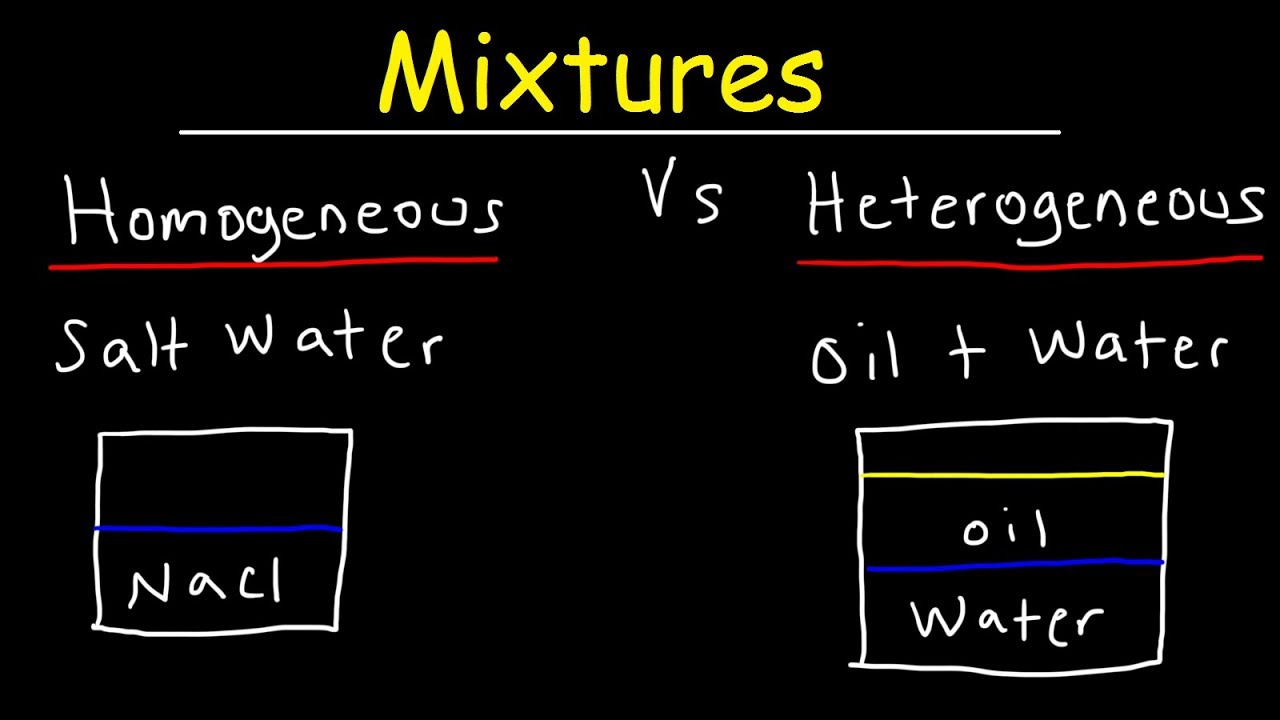
Homogenous Mixture Definition A homogenous mixture is the type of mixture in which the composition of the solute is uniform throughout the mixture The homogenous mixture can be defined by the fact that the division of the mixture into two equal halves results in the distribution of the same amount of solute suspended in both of the halvesA material composed of two or more substances is a mixture In a mixture, the individual substances maintain their chemical identities Many mixtures are obvious combinations of two or more substances, such as a mixture of sand and water Such mixtures are called heterogeneous Eg wine, beer, gelatin, water and alcohol Heterogeneous Mixtures Unlike homogeneous mixtures, in these it is very easy to identify, even to the naked eye, which are the different components that make them up This makes it much easier to separate these mixtures Eg water and oil, water and sand

Interesting Chemistry Difference Between Homogeneous And Heterogeneous Mixture Chemical Chemistrynotes Chemistry Facebook
What is an example of a heterogeneous mixture
What is an example of a heterogeneous mixture-Mixtures containing physically distinct parts and nonuniform compositions that can be seen under a microscope are called heterogeneous mixtures Example of this is sodium chloride and iron filings Was this answer helpful? A heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture Vegetable soup is a heterogeneous mixture Any given spoonful of soup will contain varying amounts of the different vegetables and other components of the soup



Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime
⭐️ Chemistry » Why would sand and water be an example of a heterogeneous mixture? In this animated lecture, I will teach you about 10 examples of homogeneous mixtures and 10 examples of heterogeneous mixtures, the meaning of homogeneous, tA mixture consists of at least two substances that are brought together in the same receptacle Here are some examples Sand and water Oil and vinegar Air and water vapor Flour, water and salt etc 2) Example of a heterogeneous mixture Before defining a heterogeneous mixture, we will see an example that will help to understand more
Most of the mixtures occurring in nature are heterogeneous For example, the soil is a mixture of hundreds of elements and compounds Its composition changes from place to place Some other examples of the heterogeneous mixture are – rocks, a mixture of kerosene and water, rice and pulses, etc Here are 10 examples of heterogeneous mixtures Cereal in milk is a great example of a heterogeneous mixture It consists of a solid cereal in liquid milk Oil and water form a heterogeneous mixture Orange juice with pulp is a heterogeneous mixture The components are unevenly distributed, plus For example, while taking a test, the test will only have content from one class Also, there is a time slot set specifically for chemistry and another for English this being an example of when these components in our "mixture" (English and chemistry) are not uniformly distributed
5 Examples Of Heterogeneous Mixtures For Chemistry Class Mixtures Heterogenous And Homogenous The term mixture is applied to two or more substances combined together, creating Terms To Describe Mixtures Solutions with identifiable particles are heterogenous solutions It is possible toHomogeneous and Heterogeneous Mixtures Examples, Classification of Matter, Chemistry Homogeneous and Heterogeneous Mixtures Examples, Classification of Matter, Chemistry Watch laterA heterogeneous mixture is simply any mixture that is not uniform in composition Two immiscible liquids form heterogeneous mixtures In given examples, water & lemon juice, water & alcohol, water & sugar result in uniform composition forming homogeneous mixture ∴ Water and stone is a heterogeneous mixture




Interesting Chemistry Difference Between Homogeneous And Heterogeneous Mixture Chemical Chemistrynotes Chemistry Facebook
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples
A mixture is a substance which consist of two or more elements or compounds not chemically combined together For Example Air is a mixture of gases like oxygen,nitrogen,argon,carbon dioxide etc Gun Powder is a mixture of potassium nitrate,sulphur,charcoal Brass is a mixture of copper and zinc The various substance present in a mixture Ans Following are the 10 different examples of heterogeneous mixtures 1) Sand amd water 2) Oil and water 3) Ice cubes in Cola 4) Applr juice with pulp in it 5) Chicken noodle soup 6) Salad 7) Tomato ketchup 8) Chocolate chips 9) Pizza 10) Sandwich To learn more about homogeneous and heterogeneous mixtures, watch this lecture till the endHomogeneous mixture Heterogeneous mixture 1) These are called as solutions These are called as suspensions/colloids 2) Substances are Uniformly distributed These substances are Unevenly distributed 3) These are not visible to the naked eye, but visible through the microscope




Heterogeneous Mixture High Res Stock Images Shutterstock



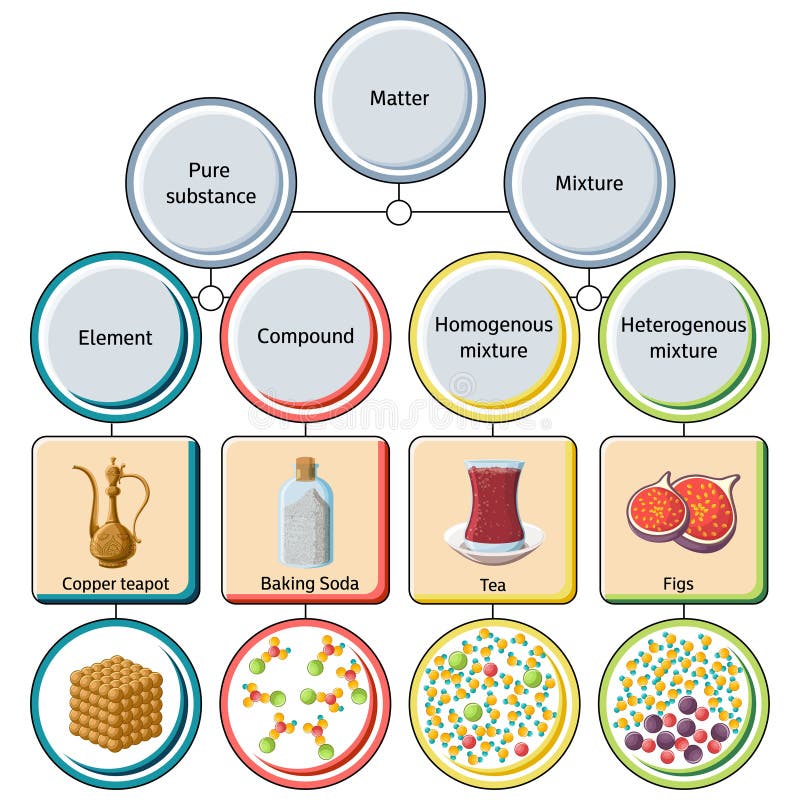
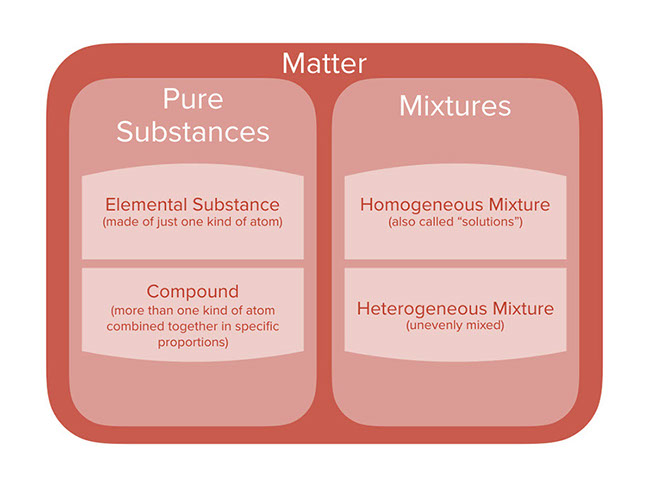
Pure Substances And Mixtures Bioprofe
Homogeneous and heterogeneous mixtures In chemistry, if the volume of a homogeneous suspension is divided in half, the same amount of material is suspended in both halves of the substance An example of a homogeneous mixture is air In physical chemistry and materials science this refers to substances and mixtures which are in a single phaseSmog is another example of a heterogeneous mixture This murky collection of pollutants can be a mixture of water and contaminants from burning gasoline or plastics mixed with nitric oxide derivatives and ozoneHomogeneous and Heterogeneous Mixtures 1 A homogeneous mixture is a solid, liquid, or gaseous mixture that has the same proportions of its components throughout any given sample In chemistry, a mixture is a material system made up of two or more different substances which are mixed but are not combined chemically




Homogeneous And Heterogeneous Mixtures Activities



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
100 heterogeneous mixture stock photos, vectors, and illustrations are available royaltyfree See heterogeneous mixture stock video clips of 1 Try these curated collections Search for "heterogeneous mixture" in these categories Examples of Heterogeneous Mixtures Concrete is a heterogeneous mixture of an aggregate cement, and water Sugar and sand form a heterogeneous mixture If you look closely, you can identify tiny sugar crystals and particles of Ice cubes in cola form a heterogeneous mixture The ice and soda are" i make a model that uses the periodic table to predict behavior and illustrate how through fission, fusion and
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



10 Heterogeneous And Homogeneous Mixtures



Properties Of Mixtures Aca Grade 8 Science
Milk Milk is heterogenous meaning made of different parts The mixture of milk has distinct parts that do not form even temporary bonds or electro static attractions as in a solution Milk is made of water, calcium, various other minerals such as magnesium, and fat molecules The fat molecules are suspended in the mixture, the random motion of the molecules ( BrownianSelect all that apply Chemical bonds are formed between the sand and water The mixture does not have a uniform appearance The mixture does not maintain a uniform composition Examples of homogeneous mixtures include Salty water — a mixture of salt and water Ruby — a mixture of Al 2 O 3 and Cr 2 O 3 Gasoline — a mixture of various hydrocarbons Brass — a mixture of Cu and Zn Air without clouds — a mixture of various gases Heterogeneous mixtures contain two or more components that can be seen, which can



Elements Compounds And Mixtures Course Hero




Types Of Mixtures In Terms Of Homogeneity Science Online
Salad A salad with lettuce, cheese, seeds, tomatoes, broccoli, and other vegetables is an example of a heterogeneous mixture Each different piece of the salad can be separated into different parts with minimal effort soil Soil is an example of a heterogeneous mixture Eg wine, gelatin, beer, coffee with milk Examples of homogeneous mixtures Wine this substance, which contains water, sugar, yeast and fruits that are mixed uniformly is another example of homogeneous mixturesExamples of homogeneous mixture A glass of lemonade (mixture of water, lemon juice, sugar, salt) is a homogeneous mixture because the dissolved sugar, salt, and lemon juice are evenly distributed throughout the entire sample You can't easily separate the lemon juice from the water;



Q Tbn And9gctjuuyeomalljsdlz1u 5uxw8ihdx0o5wibwkoouvfqqtwvrcm3 Usqp Cau




Ways To Separate Mixtures Definition Types Homogeneous Heterogeneous Mixture Eschool
An example of the latter would be a mixture of water, octane, and silicone greaseHeterogeneous solids, liquids, and gases may be made homogeneous by melting, stirring, or by allowing time to pass for diffusion to In a homogeneous mixture, on the other hand, any sample that you examine will have exactly the same composition as any other sample Within chemistry, the most common type of homogeneous mixture is a solution which is one substance dissolved completely within another Think of a solution of pure sugar dissolved in pure water Even a homogeneous mixture becomes heterogeneous if the sample size is small enough For example, blood is a homogeneous mixture, but



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




What Is A Homogeneous Mixture Definition And Examples
That means your glass of ice water, with ice cubes floating in it, is a heterogeneous mixture of homogeneous mixtures Homogeneous mixtures cannot be expressed as chemical formulas To describe any homogeneous mixture accurately, you need to name the components and tell their proportion or ratio within the mixture Heterogeneous mixtures Milk, salt and pepper, smog, chocolate chip cookie, oil and water Solutions we might use everyday Gatorade, Apple Juice, most wines and liquor, liquid detergent, coffee etc A homogeneous mixture is defined as the mixture in which solute particles are evenly distributed into a solventHomogeneous Sugar mixed in water is the most common example of a homogeneous mixture Homogeneous mixtures can be defined as the mixtures which possess the same properties and combination throughout their mass Heterogeneous A mixture of sand mixed with salt is an example of a heterogeneous mixture Heterogeneous mixtures possess different



3




Mixtures Ppt Download
Examples of Heterogeneous Mixtures Scientists recognize two types of heterogeneous mixtures; Homogenous Mixtures A homogeneous mixture is a mixture in which the composition is uniform throughout the mixture The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample Often it is easy to confuse a homogeneous mixture with a pure substance because they are bothA heterogeneous mixture has a clear identifying property where one can see various different components of the mixture It is a reaction system where the products and the reactants are found in two or more phases Some examples of a heterogeneous mixture are pizza, cookies, rocks, etc




Pure Substances Elements Compounds Homogenous Heterogenous Mixture Examples And Problems Youtube




Homogeneous Mixture Definition Lesson For Kids Video Lesson Transcript Study Com
Get an answer to your question "An example of a heterogeneous mixture is 1 soil 2 sugar 3 carbon monoxide 4 carbon dioxide " in 📙 Chemistry if there is no answer or all answers are wrong, use a search bar and try to find the answer among similar questions Mercury is an example of____ a a compound b an element c a homogeneous mixture d a heterogeneous mixture Mercury is a chemical element with the symbol Hg and atomic number 80 A chocolate chip cookie is an example of _____ a a pure substance b an element c a homogeneous mixture In chemistry, a heterogeneous mixture consists of either or both of a) multiple states of matter or b) hydrophilic and hydrophobic substances in one mixture;




Difference Between Mixtures And Compounds In Tabular Form




Homogeneous Mixture Example
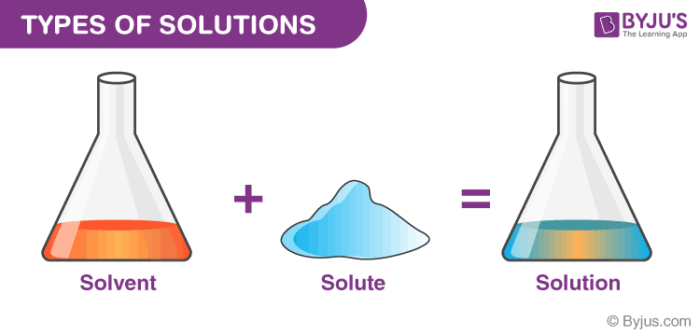
A heterogeneous mixture is a type of mixture that allows the components to be seen as two or more phases are present A mixture is an example of water Water is a homogeneous mixture of nitrogen, oxygen and smaller amounts of other compounds in the gaseous materialsA chemical mixture combines two substances that maintain their own properties when combined Heterogeneous mixtures are made up of a nonuniform composition, while homogeneous mixtures are made up of a uniform composition For example, water and sand is a heterogeneous mixture — you can easily separate the sand from the waterIn chemistry a solution is actually a type of mixture A solution is a mixture that is the same or uniform throughout Think of the example of salt water This is also called a "homogenous mixture" A mixture that is not a solution is not uniform throughout Think of the example of sand in water This is also called a "heterogeneous mixture"




Homogeneous Mixture Example Food



Inorganic Research Tweet
An example of a heterogenous mixture would be A Whipped Cream B Water C Hydrogen D Oxygen Answers 1 Show answers Another question on Chemistry Chemistry, 1950 Where do all the different elements come from?12 Examples of Common Mixtures Sea water a mixture of water and various salts Crude oil a mixture of organic compounds mainly hydrocarbons Gasoline a mixture of light hydrocarbons and performance additives Gunpowder a mixture of potassium nitrate, sulfur and carbon Dry ( air a mixture of nitrogen, oxygen, argon, carbon dioxideThey are known as suspensions and colloids Suspensions are mixtures containing particles that settle




Homogeneous Heterogeneous Mixture Definition Examples Selftution




A Solution Is An Example Of A A Homogenous Mixture Chegg Com



1




Matter And Change Mrs Coyle Chemistry A Classification




This Presentation Discusses Homogeneous And Heterogeneous Mixtures Provides Examples Explains How A Particle Di Heterogeneous Mixture Pure Products Mixtures



Is Sugar A Homogeneous Or Heterogeneous Mixture Chemistry For Neet




Heterogeneous Mixture Properties And Examples Udemy Blog



Lesson Categories Of Chemicals And Mixtures



1



Types Of Solutions Different Types Homogeneous Heterogeneous Solution With Videos



What Is A Mixture Definition Types Properties And Examples




Mixture Archives Chemistry Qna




Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime




1 A Combination Of Sand Salt And Water Is An Chegg Com




Heterogeneous Mixture Lesson For Kids Definition Examples Video Lesson Transcript Study Com




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Mixture
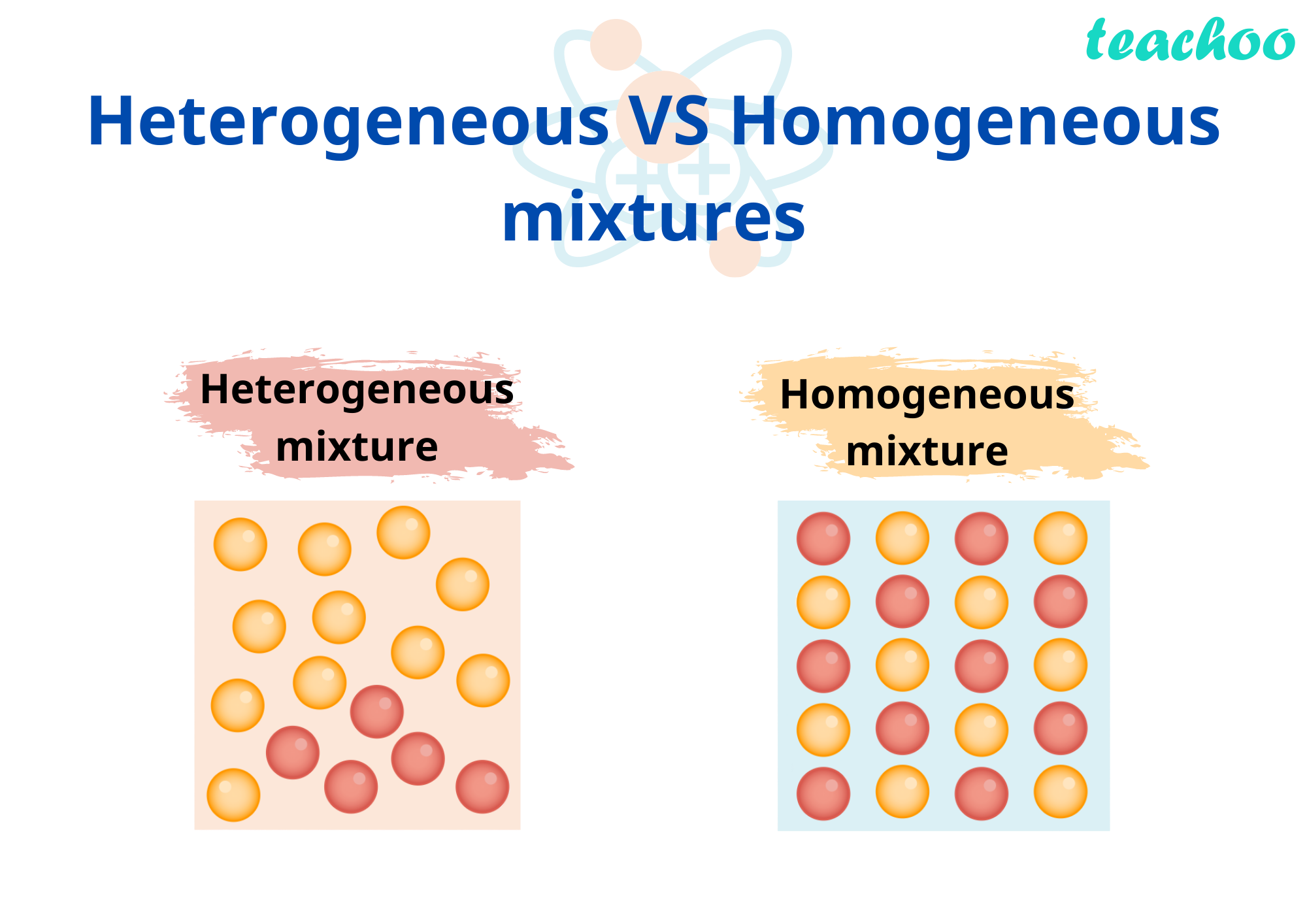



Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo




Classify Each Example As An Element A Compound A Chegg Com




What Is A Suspension Introduction Properties Videos Solved Questions
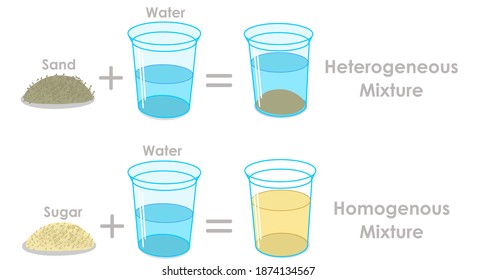



Mixtures Images Stock Photos Vectors Shutterstock
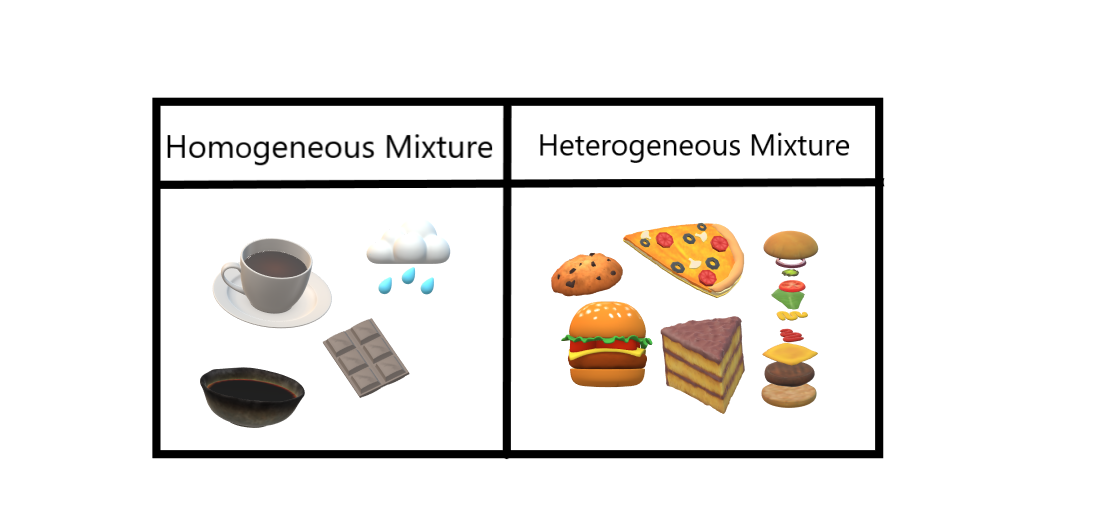



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




What Are Mixtures Definition Overview Expii




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Www Austincc Edu Mohan Documents 04 Worked Examples Pdf




Ib Chemistry Topics For Test 1 General Definitions Mole Homogeneous Mixture Heterogeneous Mixture Pure Substances Compounds Chemical Ppt Download




Mixtures And Solutions Cpd Rsc Education




Homogeneous Mixture Definition Examples Tutors Com




What Is A Homogeneous Mixture Definition And Examples
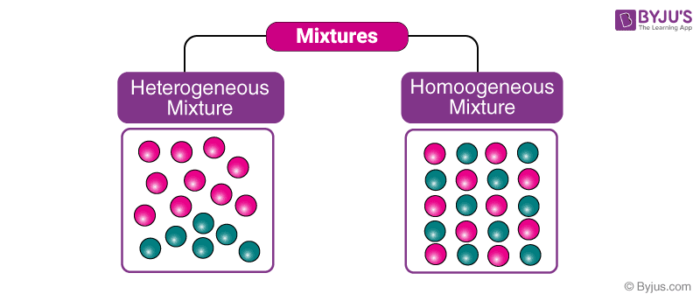



Heterogeneous And Homogeneous Mixture Differences Videos Examples




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube




5 Examples Of Heterogeneous Mixtures For Chemistry Class Science Trends




Notes On Types Of Substances Kerala Board Class 8 Chemistry




List The Points Of Differences Between Homogeneous And Heterogeneous Mixtures




Homogeneous Mixture Examples In Kitchen




Compound Vs Mixture Difference And Comparison Diffen
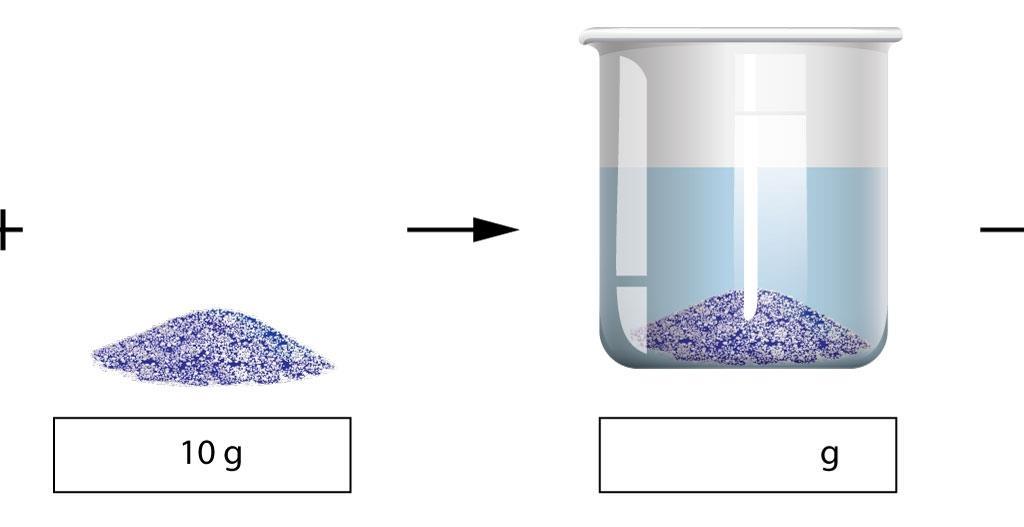



Mixtures And Solutions Cpd Rsc Education



Chem4kids Com Matter Solutions




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube




Homogeneous Mixture Definition Examples Tutors Com
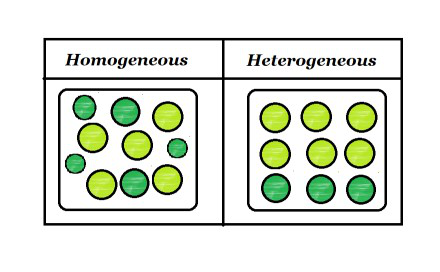



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




Chemistry Seperation Techniques Flashcards Quizlet
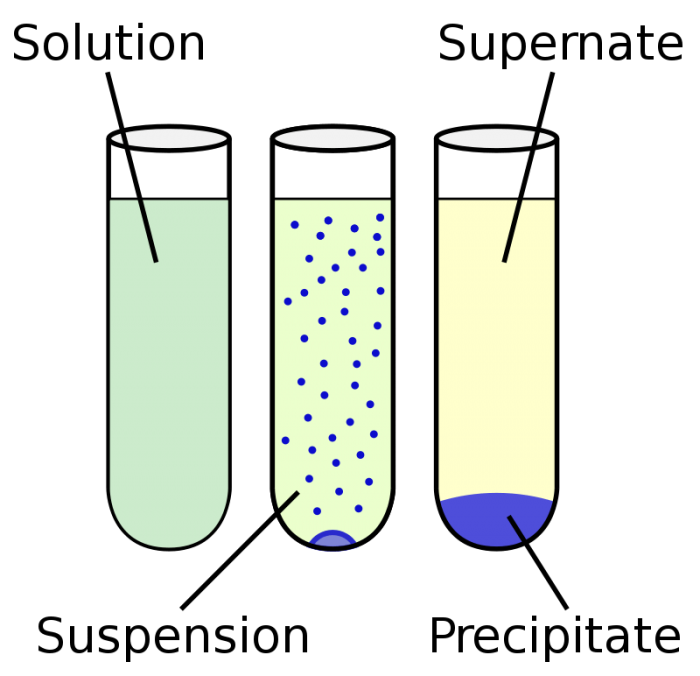



5 Examples Of Heterogeneous Mixtures For Chemistry Class Science Trends



Mixture




Examples Of Heterogeneous Mixtures Types Made Simple
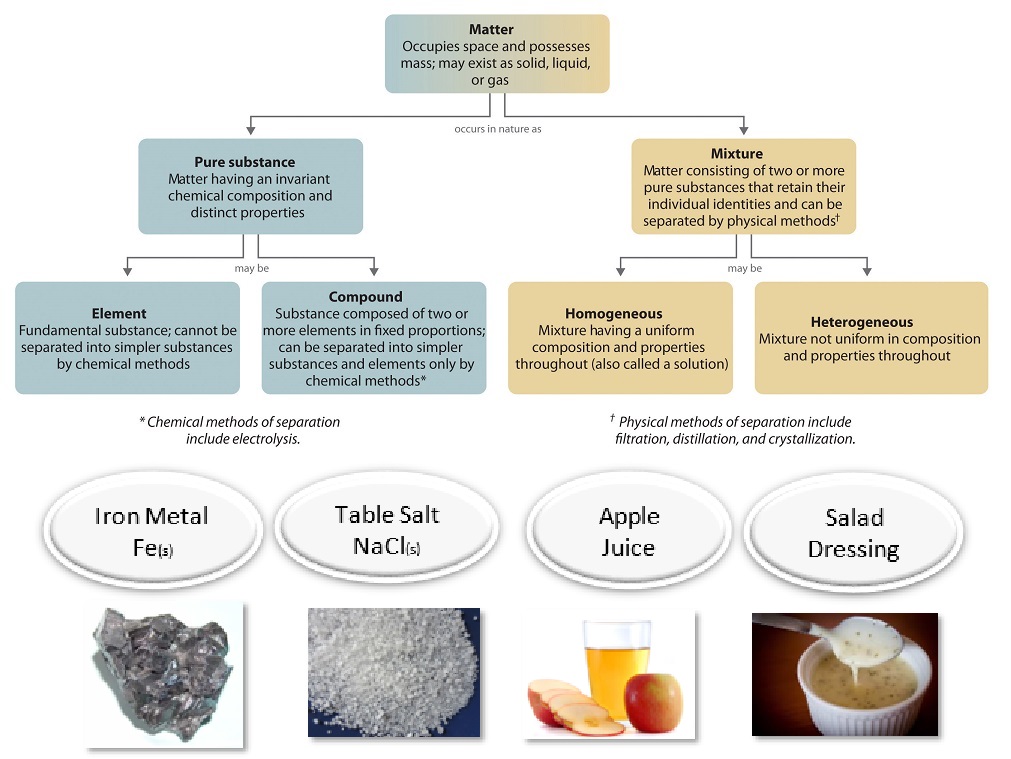



3 4 Classifying Matter According To Its Composition Chemistry Libretexts




Heterogeneous Mixture High Res Stock Images Shutterstock




5 Examples Of Homogeneous Mixture For Chemistry Class Science Trends




Homogeneous Mixture Definition Examples Tutors Com



Mixture




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Mixture Wikiwand



Homogeneous Heterogeneous Mixture Definition Examples Selftution




Heterogeneous Mixture Definition Examples Video Lesson Transcript Study Com




Chapter 1 Br Section A Br Some Basic Definitions




Teks Describe Heterogeneous And Homogeneous Mixtures 6d Explain The Similarities And Differences Between Heterogeneous And Homogenous Mixtures 6e Ppt Download



Homogeneous Mixture Experiment Qs Study



Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube




Classification Of Matter Chemistrygod




Solutions Mixture Solution




Mixture Homogeneous And Heterogeneous Mixtures Ck 12 Foundation




Examples Of Homogeneous Mixtures Solid Liquid And Gas




Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Sorting Cards




Question Video Characteristics Of Heterogeneous Mixtures Nagwa




Classification Of Matters With Examples Online Chemistry Tutorials




Let S Talk Chemistry All About Mixtures



Sxxv4y Ycx9hem




10 Examples Of Mixtures




Homogeneous Mixture Definition Examples Tutors Com




Homogeneous And Heterogeneous Mixture Heterogeneous Mixture Homogeneous Mixture Mixtures




Homogeneous Mixture Experiment Qs Study




Pure Substances And Mixtures 1 Pure And Mixed




What Is A Heterogeneous Mixture Definition And Examples



Examples Of Solution Mixtures




Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo
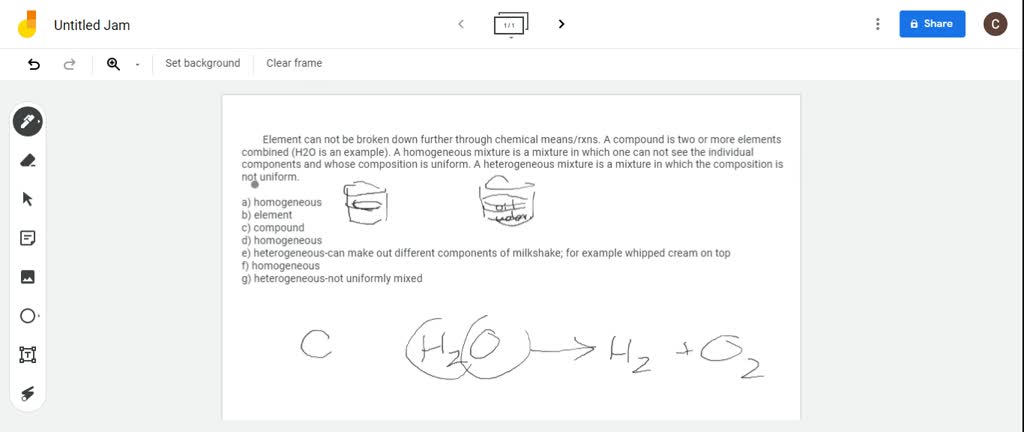



Solved 1 A Classify Each Of The Following As An Element A Compound A Homogeneous Mixture Or A Heterogeneous Mixture
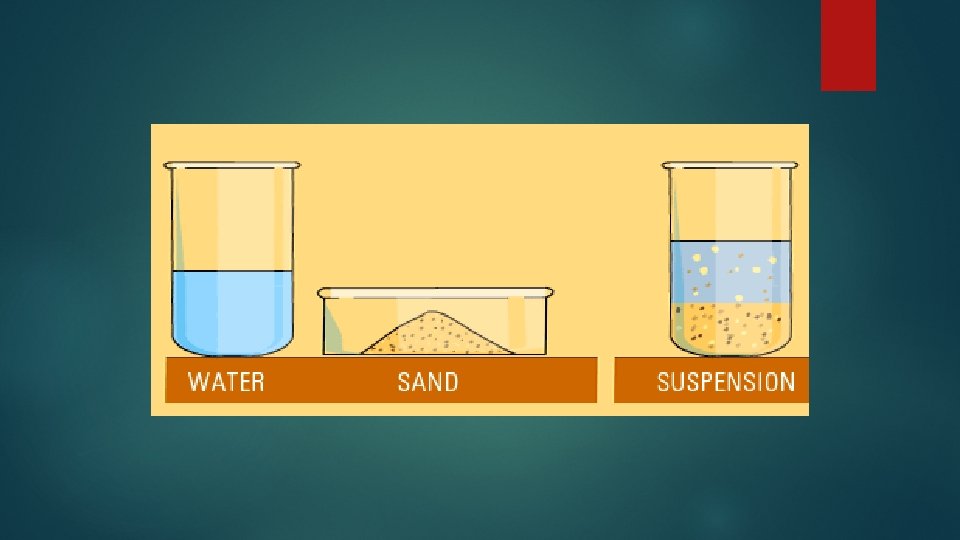



Types Of Mixtures Year 1 Science Notetaking Expectations




Chemistry Worksheet
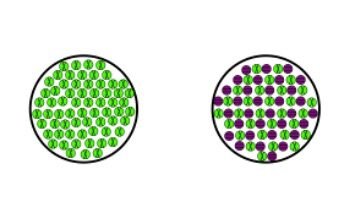



Inorganic Research Tweet



Homogeneous And Heterogeneous Mixtures
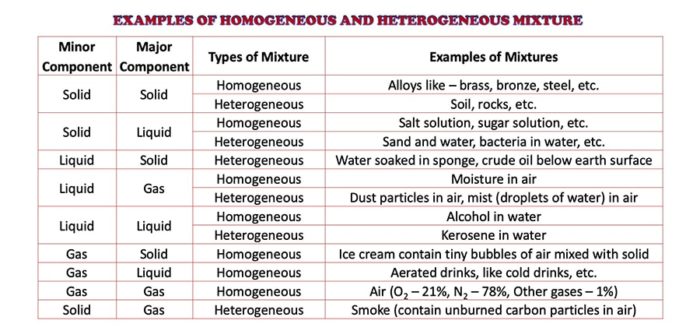



Homogeneous Heterogeneous Mixture Definition Examples Selftution



0 件のコメント:
コメントを投稿